Why 90% of Cell Therapy Contamination Cannot Be Solved by Disinfection
- Addtime: 2026-03-31 / View: 8
Introduction
As cell therapy rapidly expands across hospitals and GMP facilities, a critical issue is becoming increasingly visible:
👉 Contamination is not a rare event — it is a systemic risk.
Many facilities invest heavily in cleanroom design, validation, and disinfection protocols.
Yet after 3–6 months of operation, contamination begins to appear:
- Microbial fluctuations increase
- Repeated environmental excursions occur
- In worst cases, batch failures happen
The common response?
👉 Increase disinfection frequency
But here is the uncomfortable truth:
👉 Disinfection alone does not solve contamination.
The Misconception: Contamination is a Cleaning Problem
Most facilities treat contamination as a sanitation issue:
- Use stronger disinfectants
- Increase frequency
- Apply high-level sporicidal agents
However, contamination in cell therapy environments is rarely due to “insufficient killing.”
👉 It is due to continuous introduction and amplification of microorganisms
In other words:
👉 You are not failing to remove contamination — you are continuously generating it
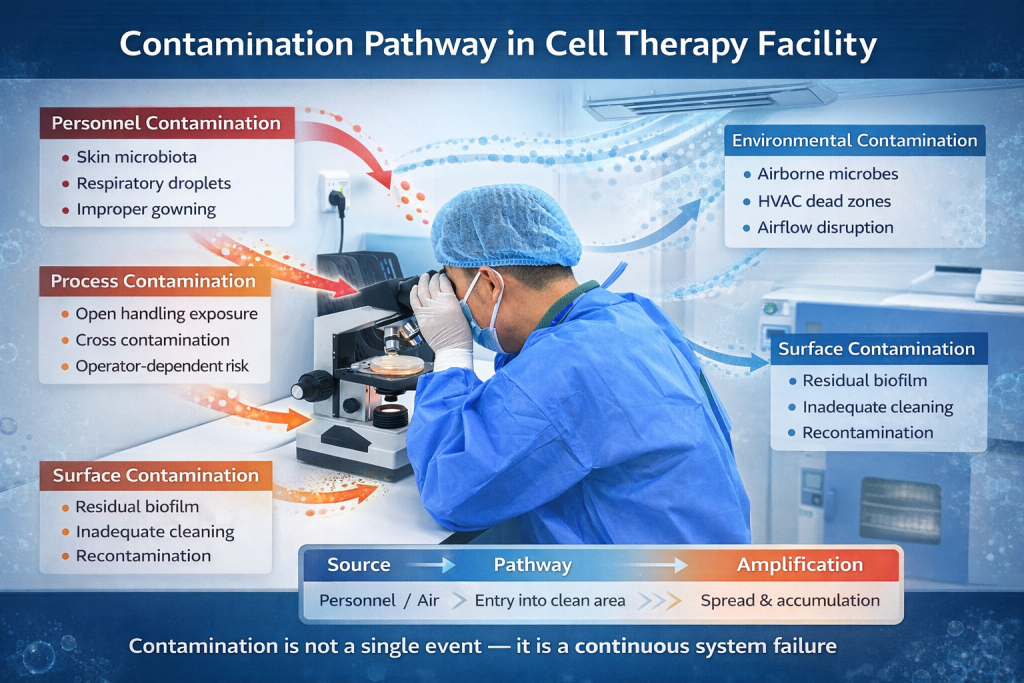
The Real Sources of Contamination
Based on extensive field experience, contamination in cell therapy facilities typically originates from three major sources:
- Facility & HVAC System Deficiencies
- Condensation caused by thermal bridges
- Dead zones in airflow
- Contaminated return air pathways
These create persistent microbial reservoirs that cannot be eliminated by surface disinfection.
👉 If the system is wrong, contamination will always come back
- Personnel & Material Flow Risks
- Cross movement between clean and non-clean zones
- Ineffective airlocks or buffer room management
- Inadequate decontamination of incoming materials
These factors continuously introduce microorganisms into controlled environments.
👉 Every movement becomes a contamination event
- Lack of Continuous Control Mechanisms
Many facilities rely on:
- Initial validation
- Periodic testing
- Reactive response after excursions
But lack:
- Trend analysis
- Early warning systems
- Real-time control strategies
👉 This leads to passive contamination management
Why Disinfection Fails as a Long-Term Solution
Disinfection is essential — but it has limitations:
- It removes existing contamination
- It does not prevent recontamination
- It does not correct system-level failures
👉 It is a corrective action, not a control strategy
Relying solely on disinfection is like:
👉 Treating symptoms while ignoring the disease
The Industry Solution: Contamination Control Strategy (CCS)
In pharmaceutical GMP environments, this challenge has already been addressed through:
👉 CCS (Contamination Control Strategy)
A CCS is a holistic system that includes:
- Facility design and airflow control
- Personnel and material flow management
- Environmental monitoring
- Risk assessment and mitigation
- Continuous improvement
👉 Disinfection becomes only the final safety layer
The Reality: Hospitals Are Not Built for This
As more hospitals begin to operate their own cell therapy facilities, a key gap emerges:
Hospitals excel in clinical practice — but not in:
- Microbial control engineering
- HVAC contamination dynamics
- Environmental risk modeling
👉 This leads to a critical mismatch:
👉 Advanced therapy + insufficient contamination control capability
A New Approach: From Disinfection to Control
The future of cell therapy facility management is shifting:
👉 From “cleaning and disinfection”
👉 To “continuous microbial control”
This includes:
- Establishing microbial baselines
- Identifying contamination pathways
- Implementing continuous monitoring
- Applying proactive control measures
- Supporting audits and compliance
In many advanced facilities, this function is evolving into:
👉 Dedicated microbial control services or on-site support teams
Conclusion
👉 The biggest risk in cell therapy is not the technology — it is environmental instability
Without a stable and controlled environment:
- Process consistency cannot be guaranteed
- Product safety is compromised
- Operational risk increases exponentially
Final Thought
👉 Contamination is not a cleaning issue — it is a system issue
Only by addressing:
- Source control
- Process stability
- Continuous monitoring
can cell therapy facilities achieve long-term, stable operation.